Abstract
Research Article
Expression of C-type Natriuretic Peptide and its Specific Guanylyl Cyclase-Coupled Receptor in Pig Ovarian Granulosa Cells
Soo Mi Kim, Suhn Hee Kim, Kyung Woo Cho, Sun Young Kim and Sung Zoo Kim*
Published: 22 August, 2018 | Volume 2 - Issue 1 | Pages: 014-025
Background: C-type natriuretic peptide (CNP) was isolated from porcine brain and is a 22-amino acid peptide which belongs to the natriuretic peptide (NP) family. Even though this peptide shares structural similarity to other endogenous NPs including atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP) its receptor selectivity is different from other NPs. The present study was undertaken to investigate the expression of C-type natriuretic peptide (CNP) and its specific guanylyl cyclase (GC)-coupled receptor in the granulosa cells of the pig ovarian follicle.
Results: Specific 125I-[Tyr0]-CNP(1-22) binding sites were localized in the granulosa cell layer of the ovarian follicle with an apparent dissociation constant (Kd>) and a maximal binding capacity (Bmax) of 1.41±0.39 nM and 2.75±0.65 fmol/mm2 respectively. Binding of 125I-[Tyr0]-CNP(1-22) to these sites was also prevented by atrial natriuretic peptide (ANP(1-28)), brain natriuretic peptide (BNP(1-26)) and des[Gln18,Ser19,Gly20, Leu21,Gly22] ANP(4-23) (C-ANP). Production of 3’,5’-cyclic guanosine monophosphate (cGMP) by particulate GC in the granulosa cell membranes was stimulated by natriuretic peptides (NPs) with a rank order of potency of CNP(1-22)>>BNP(1-26)>ANP(1-28). HS-142-1, a selective antagonist of the two recognized GC-coupled NPRs, inhibited CNP(1-22)-stimulated cGMP production in granulosa cell membranes in a dose-dependent manner. Also mRNAs for all three recognized NPRs were detected in granulosa cells using reverse transcriptase-polymerase chain reaction (RT-PCR). Serial dilution curves of granulosa cell extracts were parallel to the standard curve of synthetic CNP.
Conclusion: These results indicate that CNP and its specific receptor are expressed in the granulosa cells of the pig ovary, and suggest that CNP may be a local autocrine and/or paracrine regulator via activation of its specific GC-coupled receptor, NPR-B.
Read Full Article HTML DOI: 10.29328/journal.icci.1001004 Cite this Article Read Full Article PDF
Keywords:
Natriuretic peptide receptor; Autoradiography; RT-PCR; Granulosa layer; Pig ovary; C-Type natriuretic peptide; Particulate guanylyl cyclase; Cgmp
References
- Sudoh T, Minamino N, Kangawa K, Matsuo H. C-type natriuretic peptide (CNP): a new member of natriuretic peptide family identified in porcine brain. Biochem Biophys Res Commun. 1990; 168: 863-870. Ref.: https://tinyurl.com/yc99nj4v
- Kojima M, Minamino N, Kangawa K, Matsuo H. Cloning and sequence analysis of a cDNA encoding a precursor for rat C-type natriuretic peptide (CNP). FEBS Lett. 1990; 276: 209-213. Ref.: https://tinyurl.com/y8yjsyzx
- Kojima Y, Tsukuda Y, Kawai Y, Tsukamoto A, Sugiura J, et al. Cloning, sequence analysis, and expression of ligninolytic phenoloxidase genes of the white-rot basidiomycete Coriolus hirsutus. J Biol Chem. 1990; 265: 15224-15230. Ref.: https://tinyurl.com/yagtu7xf
- Chinkers M, Garbers DL, Chang MS, Lowe DG, Chin HM,et al. A membrane form of guanylate cyclase is an atrial natriuretic peptide receptor. Nature. 1989; 338: 78-83. Ref.: https://tinyurl.com/yd77zv7o
- Koller KJ, Lowe DG, Bennett GL, Minamino N, Kangawa K, et al. Selective activation of the B natriuretic peptide receptor by C-type natriuretic peptide (CNP). Science. 1991; 252: 120-123. Ref.: https://tinyurl.com/yc522jkf
- Stingo AJ, Clavell AL, Heublein DM, Wei CM, Pittelkow MR, et al. Presence of C-type natriuretic peptide in cultured human endothelial cells and plasma. Am J Physiol. 1992; 263: 1318-1321. Ref.: https://tinyurl.com/y9nc7txr
- Kanai Y, Yasoda A, Mori KP, Watanabe-Takano H, Nagai-Okatani C,et al. Circulating osteocrin stimulates bone growth by limiting C-type natriuretic peptide clearance. J Clin Invest. 2017; 127: 4136-4147. Ref.: https://tinyurl.com/y9yncapr
- Suga S, Nakao K, Kishimoto I, Hosoda K, Mukoyama M,et al. Phenotype-related alteration in expression of natriuretic peptide receptors in aortic smooth muscle cells. Circ Res. 1992; 71: 34-39. Ref.: https://tinyurl.com/y7pl6jl8
- Brown J, Chen Q. Regional expression of natriuretic peptide receptors during the formation of arterial neointima in the rabbit. Circ Res. 1995; 77: 906-918. Ref.: https://tinyurl.com/yc66wk2t
- Brown J, Zuo Z. Natriuretic peptide receptors in the fetal rat. Am J Physiol. 1995; 269: 253-268. https://tinyurl.com/y9q76ywq
- Langub MC Jr, Dolgas CM, Watson RE Jr, Herman JP. The C-type natriuretic peptide receptor is the predominant natriuretic peptide receptor mRNA expressed in rat hypothalamus. J Neuroendocrinol. 1995; 7: 305-309. Ref.: https://tinyurl.com/y8ygfqzn
- McArdle CA, Olcese J, Schmidt C, Poch A, Kratzmeier M, et al. C-type natriuretic peptide (CNP) in the pituitary: is CNP an autocrine regulator of gonadotropes? Endocrinology. 1994; 135: 2794-2801. Ref.: https://tinyurl.com/y8h32gzb
- Doyle DD, Ambler SK, Upshaw-Earley J, Bastawrous A, Goings GE, et al. Type B atrial natriuretic peptide receptor in cardiac myocyte caveolae. Circ Res. 1997; 81: 86-91. Ref.: https://tinyurl.com/yaxu7ac6
- Yasoda A, Komatsu Y, Nakao K, Ogawa Y. C-type natriuretic peptide(CNP)-A novel stimulator of bone growth formed through endochondral ossification. Nihon Rinsho. 2004; 62: 77-81. Ref.: https://tinyurl.com/y9cewjnu
- Yasoda A, Komatsu Y, Chusho H, Miyazawa T, Ozasa A, et al. Overexpression of CNP in chondrocytes rescues achondroplasia through a MAPK-dependent pathway. Nat Med. 2004; 10: 80-86. Ref.: https://tinyurl.com/y96wrhwg
- Bumpus FM, Pucell AG, Daud AI, Husain A. Angiotensin II: an intraovarian regulatory peptide. Am J Med Sci. 1988; 295: 406-408. Ref.: https://tinyurl.com/ycqqd9sp
- Richards AM. The renin-angiotensin-aldosterone system and the cardiac natriuretic peptides. Heart. 1996; 76: 36-44. Ref.: https://tinyurl.com/y9r8bwaa
- Vollmar AM, Mytzka C, Arendt RM, Schulz R. Atrial natriuretic peptide in bovine corpus luteum. Endocrinology. 1988; 123: 762-767. Ref.: https://tinyurl.com/ybytm8r9
- Kim SH, Cho KW, Seul KH, Ryu H, Koh GY. Presence of immunoreactive atrial natriuretic peptide in follicular fluid, ovary and ovarian perfusates. Life Sci. 1989; 45: 1581-1589. Ref.: https://tinyurl.com/y9uftvaf
- Kim SH, Cho KW, Lim SH, Hwang YH, Ryu H, et al. Presence and release of immunoreactive atrial natriuretic peptide in granulosa cells of the pig ovarian follicle. Regul Pept. 1992; 42: 153-162. Ref.: https://tinyurl.com/y7kmhtko
- Pandey KN, Osteen KG, Inagami T. Specific receptor-mediated stimulation of progesterone secretion and cGMP accumulation by rat atrial natriuretic factor in cultured human granulosa-lutein (G-L) cells. Endocrinology. 1987; 121: 1195-1197. Ref.: https://tinyurl.com/yals9q49
- Steegers EA, Hollanders JM, Jongsma HW, Hein PR. Atrial natriuretic peptide and progesterone in ovarian follicular fluid. Gynecol Obstet Invest. 1990; 29: 185-187. Ref.: https://tinyurl.com/ybpj6446
- Musah AI, Bloch JF, Baker SL, Schrank GD. Human atrial natriuretic peptide infusion and ovarian venous progesterone secretion in Yucatan micropigs. J Reprod Fertil. 1994; 101: 109-113. Ref.: https://tinyurl.com/y7dvyvxo
- Johnson KM, Hughes FM Jr, Fong YY, Mathur RS, Williamson HO, et al. Effects of atrial natriuretic peptide on rat ovarian granulosa cell steroidogenesis in vitro. Am J Reprod Immunol. 1994; 31: 163-168. Ref.: https://tinyurl.com/yaan4o3y
- Kotani E, Usuki S, Kubo T. Effect of growth hormone releasing hormone on luteinizing hormone stimulated progestin biosynthesis in cultured rat ovarian granulosa cells. Gynecol Endocrinol. 1998; 12: 307-313. Ref.: https://tinyurl.com/y7av6fy5
- Reith ME, Kim SS, Lajtha A. Binding sites for [3H] tetracaine in synaptosomal sodium channel preparations from mouse brain. Eur J Pharmacol. 1987; 143: 171-178. Ref.: https://tinyurl.com/y93lerfz
- Piao FL, Park SH, Han JH, Cao C, Kim SZ, et al. Dendroaspis natriuretic peptide and its functions in pig ovarian granulosa cells. Regul Pept. 2004; 118: 193-198. Ref.: https://tinyurl.com/ybdfn2ya
- Brunswig B, Budnik LT, Mukhopadhyay AK. Atrial natriuretic peptide-induced stimulation of cyclic GMP formation by isolated bovine luteal cells. J Reprod Fertil. 1989; 86: 665-669. Ref.: https://tinyurl.com/y7x66drg
- Jankowski M, Reis AM, Mukaddam-Daher S, Dam TV, Farookhi R, et al. C-type natriuretic peptide and the guanylyl cyclase receptors in the rat ovary are modulated by the estrous cycle. Biol Reprod. 1997; 56: 59-66. Ref.: https://tinyurl.com/ycb6drkz
- Benfenati F, Cimino M, Agnati LF, Fuxe K. Quantitative autoradiography of central neurotransmitter receptors: methodological and statistical aspects with special reference to computer-assisted image analysis. Acta Physiol Scand. 1986; 128: 129-146. Ref.: https://tinyurl.com/ychmcvgt
- Munson PJ, Rodbard D. Ligand: A versatile computerized approach for characterization of ligand-binding systems. Anal Biochem. 1980; 107: 220-239. Ref.: https://tinyurl.com/ybj89aoq
- Cho KW, Seul KH, Kim SH, Seul KM, Koh GY. Atrial pressure, distension, and pacing frequency in ANP secretion in isolated perfused rabbit atria. Am J Physiol. 1991; 260: 39-46. Ref.: https://tinyurl.com/y9qeg7q9
- Joseph LJ, Desai KB, Mehta MN, Mathiyarasu R. Measurement of specific activity of radiolabelled antigens by a simple radioimmunoassay technique. Int J Rad Appl Instrum B. 1988; 15: 589-590. Ref.: https://tinyurl.com/ychgxppc
- Schulz S, Singh S, Bellet RA, Singh G, Tubb DJ, et al. The primary structure of a plasma membrane guanylate cyclase demonstrates diversity within this new receptor family. Cell. 1989; 58: 1155-1162. Ref.: https://tinyurl.com/ybjosdok
- Kim SH, Cho KW, Oh SH, Hwang YH, Lim SH, et al. Immunoreactive atrial natriuretic peptides in the oocyte. Comp Biochem Physiol Comp Physiol. 1993; 104: 219-223. Ref.: https://tinyurl.com/ya6nradz
- Tornell J, Carlsson B, Billig H. Atrial natriuretic peptide inhibits spontaneous rat oocyte maturation. Endocrinology. 1990; 126: 1504-1508. Ref.: https://tinyurl.com/ycx8ykmc
- Porter JG, Wang Y, Schwartz K, Arfsten A, Loffredo A, et al. Characterization of the atrial natriuretic peptide clearance receptor using a vaccinia virus expression vector. J Biol Chem. 1988; 263: 18827-18833. Ref.: https://tinyurl.com/yc6kgyyj
- Maack T, Suzuki M, Almeida FA, Nussenzveig D, Scarborough RM, et al. Physiological role of silent receptors of atrial natriuretic factor. Science. 1987; 238: 675-678. Ref.: https://tinyurl.com/y97c5m5f
- Schenk DB, Phelps MN, Porter JG, Fuller F, Cordell B, et al. Purification and subunit composition of atrial natriuretic peptide receptor. Proc Natl Acad Sci USA. 1987; 84: 1521-1525. Ref.: https://tinyurl.com/y8xd4a62
- Leitman DC, Andresen JW, Catalano RM, Waldman SA, Tuan JJ, et al. Atrial natriuretic peptide binding, cross-linking, and stimulation of cyclic GMP accumulation and particulate guanylate cyclase activity in cultured cells. J Biol Chem. 1988; 263: 3720-3728. Ref.: https://tinyurl.com/ya7w3zs9
- Thibault G, Grove KL, Deschepper CF. Reduced affinity of iodinated forms of Tyr0 C-type natriuretic peptide for rat natriuretic peptide receptor B. Mol Pharmacol. 1995; 48: 1046-1053. Ref.: https://tinyurl.com/ybkqvey4
- Morishita Y, Sano T, Kase H, Yamada K, Inagami T, et al. HS-142-1, a novel nonpeptide atrial natriuretic peptide (ANP) antagonist, blocks ANP-induced renal responses through a specific interaction with guanylyl cyclase-linked receptors. Eur J Pharmacol. 1992; 225: 203-207. Ref.: https://tinyurl.com/y8aen42e
- Morishita Y, Matsuda Y. HS-142-1; a novel polysaccharide, specifically recognizes guanylyl cyclase-containing ANP receptor. Tanpakushitsu Kakusan Koso. 1993; 38: 903-908. Ref.: https://tinyurl.com/y8ebbz2b
Figures:
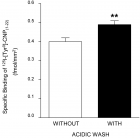
Figure 1
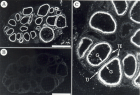
Figure 2
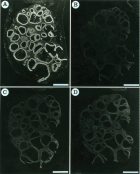
Figure 3

Figure 4

Figure 5
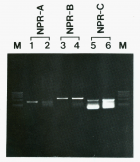
Figure 6
Similar Articles
-
Expression of C-type Natriuretic Peptide and its Specific Guanylyl Cyclase-Coupled Receptor in Pig Ovarian Granulosa CellsSoo Mi Kim,Suhn Hee Kim,Kyung Woo Cho,Sun Young Kim,Sung Zoo Kim*. Expression of C-type Natriuretic Peptide and its Specific Guanylyl Cyclase-Coupled Receptor in Pig Ovarian Granulosa Cells. . 2018 doi: 10.29328/journal.icci.1001004; 2: 014-025
-
Modulation of atrial natriuretic peptide receptors in ovarian folliculogenesisSung Zoo Kim*. Modulation of atrial natriuretic peptide receptors in ovarian folliculogenesis. . 2022 doi: 10.29328/journal.icci.1001019; 6: 001-007
Recently Viewed
-
From the Single Bacterial Cell to the Microbial Community: A Round Trip to better understand the Secrets of Complex Microbiological EcosystemsErasmo Neviani*. From the Single Bacterial Cell to the Microbial Community: A Round Trip to better understand the Secrets of Complex Microbiological Ecosystems. Int J Clin Microbiol Biochem Technol. 2024: doi: 10.29328/journal.ijcmbt.1001029; 7: 006-008
-
Renal Malakoplakia: A Diagnostic Challenge Presenting as a Subcapsular Collection despite Clinical RecoveryMohammed Amine Elafari*,Mamad Ayoub,Mohammed Amine Bibat,Amine Slaoui,Tarik Karmouni,Abdelatif Koutani,Khalid Elkhader. Renal Malakoplakia: A Diagnostic Challenge Presenting as a Subcapsular Collection despite Clinical Recovery. J Clin Med Exp Images. 2026: doi: 10.29328/journal.jcmei.1001046; 10: 032-038
-
Not Every Bladder Mass Is Malignant: A Case of Inverted Urothelial Papilloma in a Young AdultMohammed Amine Elafari*,Mamad Ayoub,Mohammed Amine Bibat,Amine Slaoui,Tarik Karmouni,Abdelatif Koutani,Khalid Elkhader. Not Every Bladder Mass Is Malignant: A Case of Inverted Urothelial Papilloma in a Young Adult. J Clin Med Exp Images. 2026: doi: 10.29328/journal.jcmei.1001043; 10: 016-019
-
Adult Bladder Exstrophy with Premalignant Changes Following Failed Reconstruction: A Case ReportMohammed Amine Elafari*,Mamad Ayoub,Mohamed Amine Zaki,Mohammed Amine Bibat,Amine Slaoui,Tarik Karmouni,Abdelatif Koutani,Khalid Elkhader. Adult Bladder Exstrophy with Premalignant Changes Following Failed Reconstruction: A Case Report. J Clin Med Exp Images. 2026: doi: 10.29328/journal.jcmei.1001044; 10: 020-023
-
Urinary Biomarkers: Is the Era of Exclusive Follow-Up Cystoscopy Coming to an End? A Mini-Review of Emerging Molecular Diagnostics and Risk-Stratified SurveillanceMohammed Amine Elafari,Mamad Ayoub,Mohammed Amine Bibat,Amine Slaoui,Tariq Karmouni,Abdelatif Koutani,Khalid Elkhader. Urinary Biomarkers: Is the Era of Exclusive Follow-Up Cystoscopy Coming to an End? A Mini-Review of Emerging Molecular Diagnostics and Risk-Stratified Surveillance. Arch Cancer Sci Ther. 2026: doi: 10.29328/journal.acst.1001050; 10: 011-015
Most Viewed
-
Physical Performance in the Overweight/Obesity Children Evaluation and RehabilitationCristina Popescu, Mircea-Sebastian Șerbănescu, Gigi Calin*, Magdalena Rodica Trăistaru. Physical Performance in the Overweight/Obesity Children Evaluation and Rehabilitation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001030; 8: 004-012
-
Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging PresentationKarthik Baburaj*, Priya Thottiyil Nair, Abeed Hussain, Vimal MV. Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging Presentation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001029; 8: 001-003
-
Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trialSathit Niramitmahapanya*,Preeyapat Chattieng,Tiersidh Nasomphan,Korbtham Sathirakul. Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trial. Ann Clin Endocrinol Metabol. 2023 doi: 10.29328/journal.acem.1001026; 7: 00-007
-
Exceptional cancer responders: A zone-to-goDaniel Gandia,Cecilia Suárez*. Exceptional cancer responders: A zone-to-go. Arch Cancer Sci Ther. 2023 doi: 10.29328/journal.acst.1001033; 7: 001-002
-
Ectopic adrenal tissue at the spermatic cordAbdallah Chaachou,Nizar Cherni,Wael Ferjaoui*,Mohamed Dridi,Samir Ghozzi. Ectopic adrenal tissue at the spermatic cord. J Clin Med Exp Images. 2022 doi: 10.29328/journal.jcmei.1001024; 6: 001-002

If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."